We’re excited to share the second of three recent research papers featuring our very own CoolLED pE-800 Illumination System.
This month, you can learn about the role of spatial biology in cancer diagnostics.
The Future of Biomarker Analysis
Cancer is a notoriously diverse and complex disease, where clinical insights benefit from understanding the tumour microenvironment – the adjacent non-tumour cells which support its survival, growth and spread.
Spatial information is crucial for this application, but analysing biomarkers from the tumour microenvironment is tricky using traditional brightfield dyes, which have limited spectral separation. For clinical assays, multiplexing fluorescent dyes isn’t practical either due to the high throughput required.
In a recent study, researchers instead looked towards a different approach, developing a chromogenic assay which utilises existing high-throughput infrastructure such as the tissue microarray for brightfield assays.
They designed a spectroscopic imaging system capable of multiplexing, and also investigated the potential of combining this with the latest developments in digital pathology.
How does it work?
As light travels through a sample, each chromogen absorbs its own spectral ‘footprint’, indicating the location and concentration of the labelled biomarker.
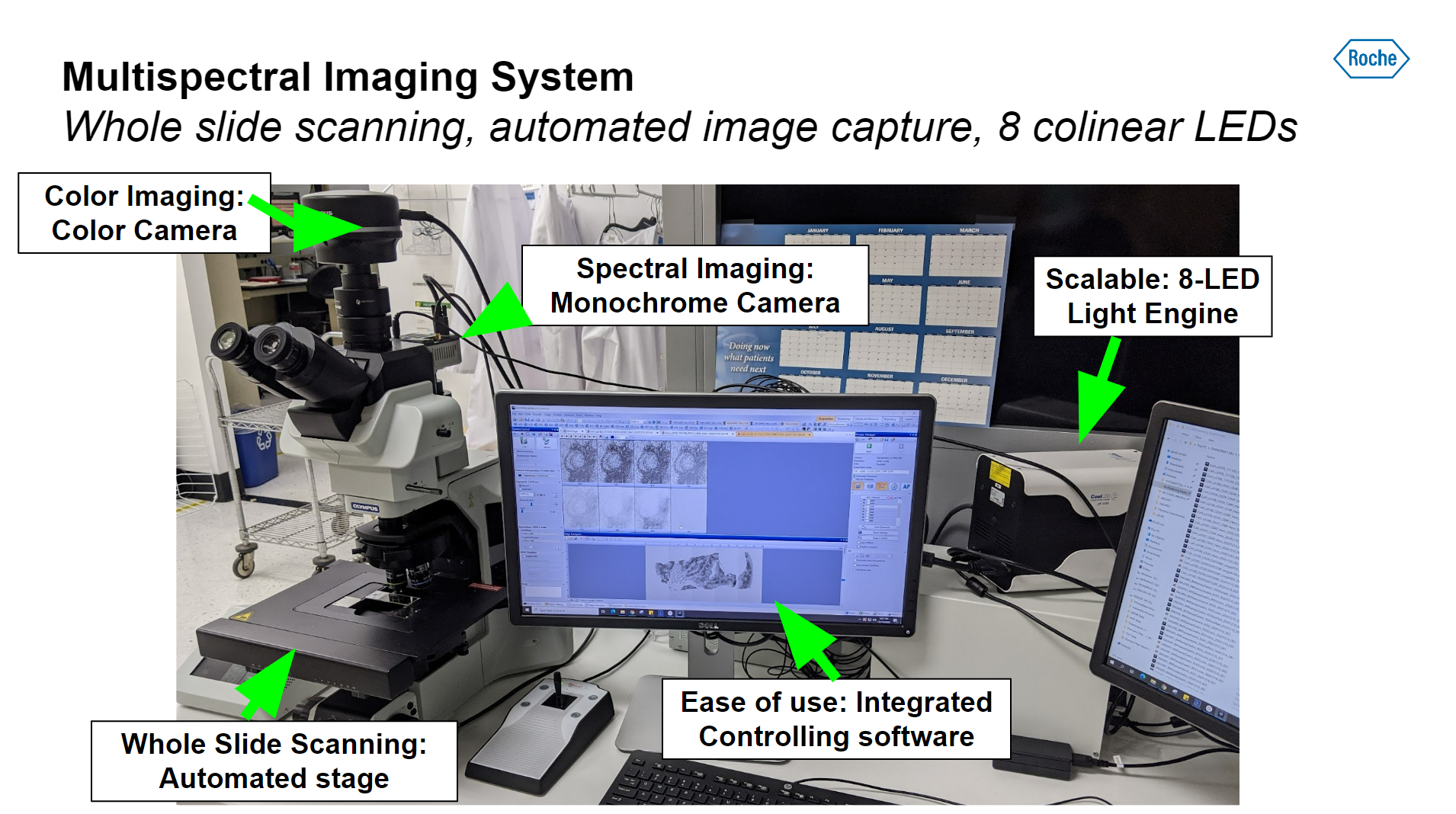
The hardware setup is based on a standard clinical upright microscope, with illumination from the 8-channel CoolLED pE-800 LED Illumination System. However, unlike the standard epi-fluorescence applications of this light source, this spectroscopic imaging technique uses the transmitted light path (allowing light to pass through the sample). The pE-800 provides several valuable features here, including:
- Eight individually controllable LED channels: when combined with matching excitation filters, eight discrete channels of narrow-band illumination are able to closely match the absorbance wavelengths of multiple chromogens.
- The ability to house excitation filters in front of each LED removes the need for a filter wheel, increasing imaging speed.
- TTL synchronisation to the camera ensures precise automated imaging over long acquisition sequences.
- The high-speed (<7 µs) TTL triggering enables faster imaging and therefore sample throughput – and time is of the essence in a clinical diagnostic setting.
Once the transmitted illumination images are reversed to show absorbance, and spectral unmixing is applied, these footprints can be visualised from multiple biomarkers in parallel – providing information-rich images ready for further digital analysis.
What was the outcome?
The platform was used to test a novel 6-marker prostate cancer assay, which delivered quantitative data on protein concentration and even spatial distribution patterns compared to healthy tissue.
Introducing novel technologies into the clinical workflow presents many challenges, but the ability to apply a spatial biology approach combined with powerful AI and other digital pathology tools provides a wealth of opportunity to understand the complexities of cancer and rise to the challenge of personalised medicine.
About the pE-800
The CoolLED pE-800 includes eight LED channels covering the major fluorophores and opsins.
The ability to have any combination of these switched on and control the intensity allows multiple fluorophores to be excited in parallel, while high-speed TTL triggering and synchronisation enables accurate and precise control of complex pulsing protocols.
This is particularly useful for neuroscience applications.

CoolLED specialises in LED microscope lighting and, since our team of four introduced the first commercially available LED microscope light source in 2006, we have led the way in designing and manufacturing cutting edge LED Illuminators for Microscopes using the latest technology.







